Giatec continues to uphold the value of bridging the gap between academic research and sustainable construction on the jobsite. Every year, we recognize civil engineering students, researchers and faculty across Canada and the U.S. with the Giatec Best Paper Award for Sustainability in Construction. Below is a summary of the 2022 winning paper submitted by Prof. Caijun Shi and his team, Effect of Temperature on CO2 Curing, Compressive Strength and Microstructure of Cement Paste. Find how you can join the list of winners and showcase your research to a global audience of industry experts while receiving a monetary award of 1,000 USD here.
Build Data Centers Faster with SmartRock® Long Range
Research Background
The increase in the concentration of atmospheric carbon dioxide (CO2) began more than a century ago with the industrial revolution in the west and continues to grow. One of the biggest concerns about rising CO2 is its contribution to climate change. Experts reported that the earth warms proportionally with increasing CO2 concentration. Therefore, many technologies and regulations have been introduced over the past decades to cut atmospheric concentrations. The direct capture of CO2 from the air has proven its efficiency in drawing down the CO2 level but concerns remain on the cost and effectiveness of storing it underground. Therefore, many scientists have suggested transporting the captured CO2 and using it as part of the curing process of concrete. The idea was to create a “sink” for CO2 within concrete, without harming or negatively affecting the strength and durability of the end product.
Prof. Caijun Shi and his team developed a pre-conditioning technology and demonstrated that CO2 curing could (1) sequestrate up to 20% CO2 permanently in stable and durable minerals (2) reduce significantly the time required for curing to several hours to reach a strength of 25MPa or higher and (3) improve the durability of concrete. However, the variability in the curing conditions at a particular time and temperature could have a significant impact on the CO2 curing process. As such, Prof. Shi’s team is extensively investigating the CO2 curing protocol with the overall goal of identifying optimal curing conditions and design mixes to maximize the benefits of CO2 curing and preserve concrete properties.
Important Findings
The CO2-cured samples exhibited about 3 times higher early-age compressive strength compared to that of water-cured samples after 2h of curing, regardless of temperature. In addition, CO2 curing improved the compressive strength more effectively at 50℃ compared to that at 5℃ and 20℃ (Fig.1). This is probably due to the higher efficiency in hydration and carbonation reactions at higher temperatures, offsetting typically the lower strength gain induced by lower curing temperature. At 20℃ and 50℃ a strength greater than 40 MPa was achieved after 12 h of CO2 curing, indicating that CO2 curing could significantly reduce the curing time. This can also suggest that 12 hours of CO2 curing could be considered close to an optimum curing time to reach a satisfying strength.
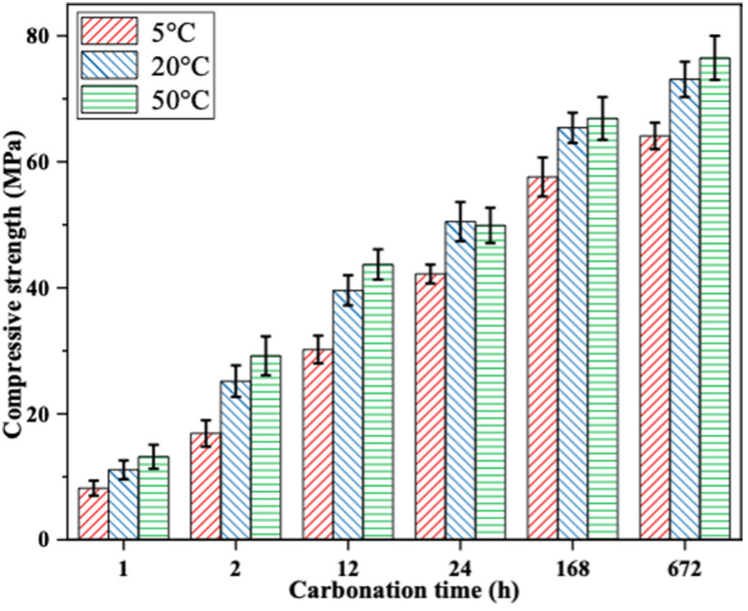
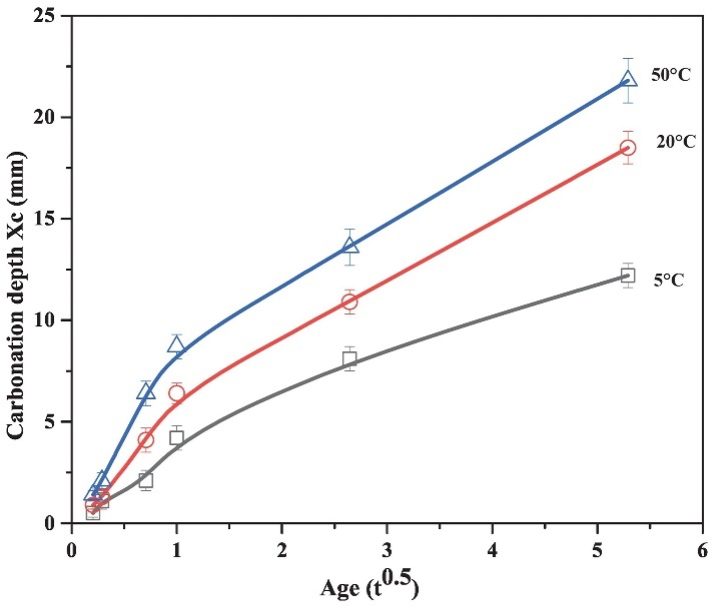
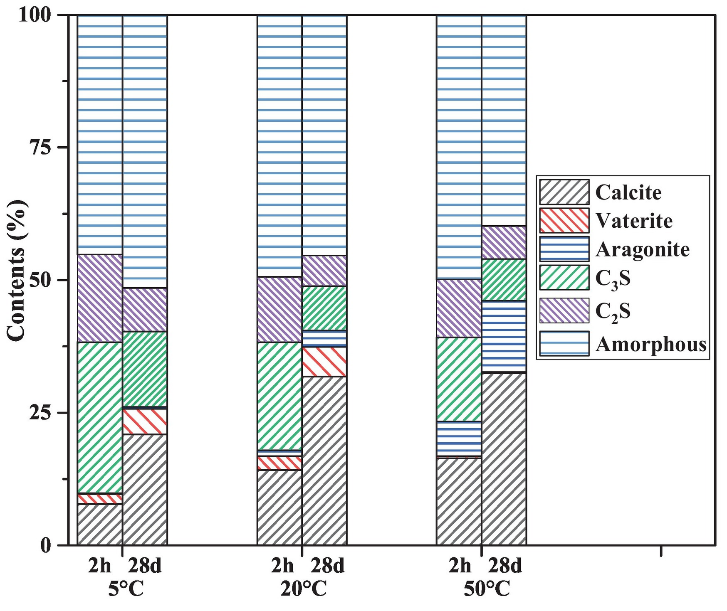
Similar to conventional curing practices, temperature significantly accelerated the carbonation kinetics (Fig. 2), improved the rate of strength development (Fig. 1), and influenced the microstructure (Fig. 3) of cement paste. This has been explained as follows: an increase in temperature leads to the evaporation of capillary water from a large number of pores and consequently promoted the diffusion of CO2 in the matrix. However, lack of water slows carbonation. This explains the slower enhancement rate of the above properties with an extended CO2 curing time. The layer of calcium carbonates and silica gel formed on the anhydrous cement particles could also have contributed to this result since it could prevent more water and CO2 from reaching the cement particles and consequently halt the hydration/carbonation reaction.
For more details, readers can refer to the paper Effect of Temperature on CO2 Curing, Compressive Strength, and Microstructure of Cement Paste published in Cement and Concrete Research. This study was conducted by Prof. Caijun Shi and his team and is freely available online with no additional restrictions thanks to the open access agreement between OsloMet – Oslo Metropolitan University and Elsevier.
Bao Lu, Sarra Drissi, Jianhui Liu, Xiang Hu, Baixin Song, Caijun Shi
Lead author, Caijun Shi, is a chair professor at Hunan University in China and an affiliate professor at the University of British Columbia. He can be contacted at cshi@hnu.edu.cn







