Corrosion is a natural process that occurs when a structure is exposed to elements like CO2 or chloride, which can penetrate the concrete all the way to the steel reinforcement. This can have serious durability and safety consequences, which is why it is important to monitor corrosion using an accurate and trusted method. The half-cell potential test is the only corrosion monitoring technique standardized in ASTM C876 – 15: Standard Test Method for Corrosion Potentials of Uncoated Reinforcing Steel in Concrete. It is used to determine the probability of corrosion within the rebar in reinforced concrete structures. This blog dives into the specifics of concrete corrosion, the half-cell potential measurement for testing concrete corrosion, and the ways in which the data from a half-cell potential device can be interpreted.
Detect the Rate of Rebar Corrosion in Seconds
The Basics of Concrete Corrosion
In reinforced concrete structures, there is a natural protective film that forms on the surface and prevents the bar from corroding. With time, chlorides (from de-icing salts or marine exposure) and/or CO2 penetrate the concrete and breakdown that protective layer. Chlorides destabilize the passive film leading to its localized breakdown, while CO2 lowers the pH of the concrete below the level of stability of the passive film. In the presence of oxygen and water, an electrochemical reaction initiates the process of corrosion.
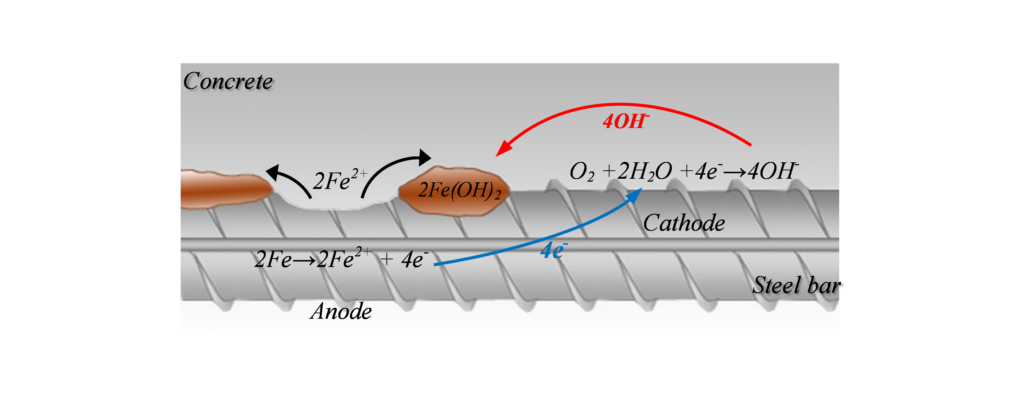
Corrosion can be illustrated as shown in Figure 1, where the metal (rebar) reacts in the solution (available in the concrete pores) and gives away electrons from the anode (where oxidization occurs) to the cathode (where reduction occurs). The positive ions formed at the surface of the anode will react and create corrosion by-products. This electrochemical reaction creates a potential difference, and consequently a corrosion current, between the anodic and cathodic areas at the surface of the steel reinforcement. This current, or the potential distribution on the reinforcement surface, is of interest for half-cell potential measurement.
Half-Cell Potential Measurement
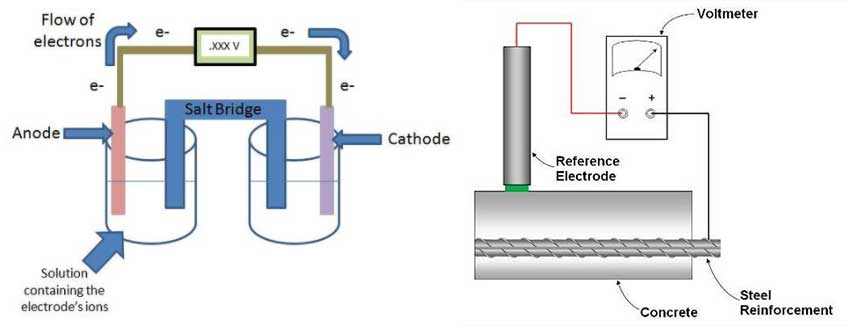
The schematic in Figure 2a represents a cell where each side is referred to as a half-cell. Each half-cell is represented by an electrode in a solution (electrolyte) and both half-cells are connected together. Since one of the electrodes has a higher tendency to corrode compared to the other, that electrode (anode) will oxidize and in turn will donate electrons.
To keep the system in equilibrium and balance the charges in the electrolytes, there will be an exchange of ions through the salt bridge. The voltmeter will measure the potential difference (voltage) between both electrodes, which indicates the rate of dissolution of the anode.
Interested in detecting corrosion in reinforced concrete? Learn more here!
To apply this concept to concrete and to interpret the results of corrosion potential, a reference electrode with a known potential is needed. Typically, for reinforced concrete applications, a copper/copper sulfate electrode (Cu/CuSO4) or silver/silver chloride electrode (Ag/AgCl) is used for the half-cell reference. This reference electrode is connected to the other half-cell represented by the embedded rebar (Figure 2b). By connecting that reference electrode to the reinforcing steel and placing the reference electrode on the surface of the concrete, it is possible to measure the potential difference between the two half-cells.
ASTM C876 provides a guideline on how this measurement can be undertaken, and the relationship between the measured potential values and the corrosion probability. Interpretation of the result is qualitative and is based on the copper sulfate electrode (CSE). Table 1 shows the general interpretation guideline proposed by ASTM, where the measured potential ranges are categorized in three categories; more than 90% chance, less than 10% chance or an uncertain chance of corrosion.
Table 1: Relationship between the potential values and corrosion probability
(adapted from ASTM C876)
| Measured Potential(mV CSE) | Probability of steelcorrosion activity |
|---|---|
>-200 | Less than 10% |
-200 to -350 | Uncertain |
<-350 | More than 90% |
Interpreting Half-Cell Potential Data
At first glance, this test method seems very simple and comprises of the following steps:
- Identify rebar location
- Make a connection with the reinforcement (more than one connection can be required if there is a discontinuity between reinforcements)
- Prepare concrete surface through wetting
Measurements are quick as potential values only take a few seconds to stabilize before the next measurement can be taken. However, there are important limitations in terms of data interpretation that need to be taken into consideration.
The effect of the concrete condition (dry or wet), presence of chloride, absence of oxygen at the rebar surface (due to saturation), cover thickness, concrete resistivity, and temperature are all factors that can influence the results by shifting their potential reading towards a more positive or negative value as shown in Table 2. This can make the data interpretation challenging when using the guidelines given in the ASTM C876 (Table 1), especially around the uncertain measurement ranges.
Table 2: Typical ranges of half-cell potentials of rebar in concrete (adapted from RILEM TC-154, 2003)
| Conditions | Potential values (mV/CSE*) |
|---|---|
| Humid, chloride free concrete | -200 to +100 |
| Wet, chloride contaminated concrete | -600 to -400 |
| Water saturated concrete without oxygen | -1000 to -900 |
| Humid, carbonated concrete | -400 to +100 |
| Dry, carbonated concrete | 0 to +200 |
| Dry concrete | 0 to +200 |
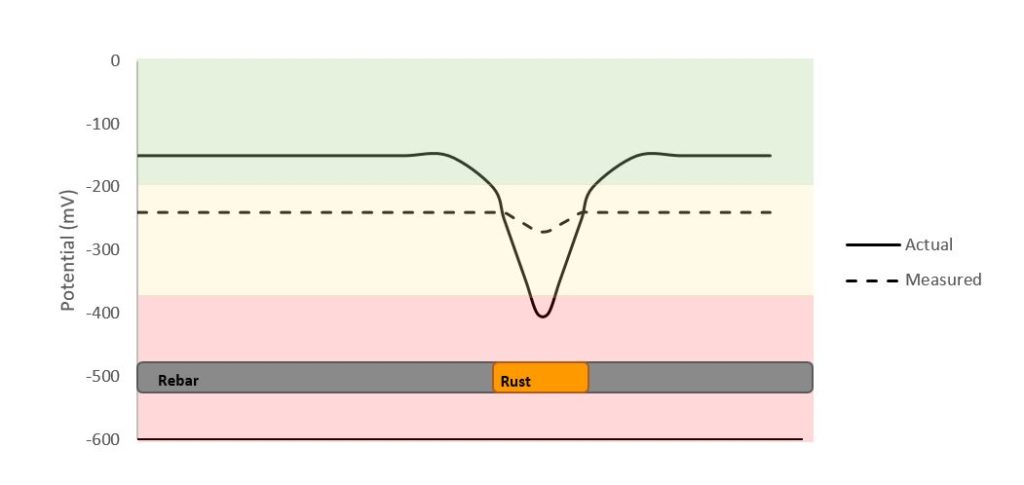
In addition, half-cell measurement is considered to be a zonal measurement as it will take an average potential measurement of the surroundings. An example is illustrated in Figure 3, where the measured potential will show sort of an average over a certain distance and where the actual location of the corroded bar can be challenging to distinguish, even with corrosion potential mapping.
Nevertheless, this technique is being widely used since it is the only corrosion monitoring technique standardized by ASTM. Giatec’s iCOR® device which measures corrosion rate and concrete resistivity, can also calculate half-cell potential. It uses a silver/silver chloride electrode, making this NDT device more stable and accurate than those half-cell devices, which use a copper/copper sulfate electrode. It can be very useful in doing a quick assessment and identifying the regions where there might be a relatively higher corrosion activity. It remains that the corrosion potential technique’s output is qualitative, as it provides information only on the chance of corrosion activity, not quantitative information such as the rate at which the rebar is corroding; this is more useful information when it comes to determining the plan of action for repair or corrosion mitigation.
Related Readings: Obtaining Effective Half-Cell Potential Measurements in Reinforced Concrete Structures
Half-Cell Potential Measurements – Potential Mapping on Reinforced Concrete Structures
**Editor’s Note: This post was originally published in May 2018 and has been updated for accuracy and comprehensiveness in November 2025.









